Relating Structural and Microstructural Evolution to the Reactivity of Cellulose and Lignin during Alkaline Thermal Treatment wi

Effect of Impurities on the Decarbonization of Calcium Carbonate Using Aqueous Sodium Hydroxide | ACS Sustainable Chemistry & Engineering

Comparative Study of Calcium Carbonates and Calcium Phosphates Precipitation in Model Systems Mimicking the Inorganic Environment for Biomineralization | Crystal Growth & Design

Analysis of Operation Conditions of Ca(OH)2 Entrained Carbonator Reactors for CO2 Capture in Backup Power Plants | ACS Omega

PDF) Electrochemical Splitting of Calcium Carbonate to Increase Solution Alkalinity: Implications for Mitigation of Carbon Dioxide and Ocean Acidity

Effect of Impurities on the Decarbonization of Calcium Carbonate Using Aqueous Sodium Hydroxide | ACS Sustainable Chemistry & Engineering

Calcium-Looping Performance of Biomineralized CaCO3 for CO2 Capture and Thermochemical Energy Storage | Industrial & Engineering Chemistry Research
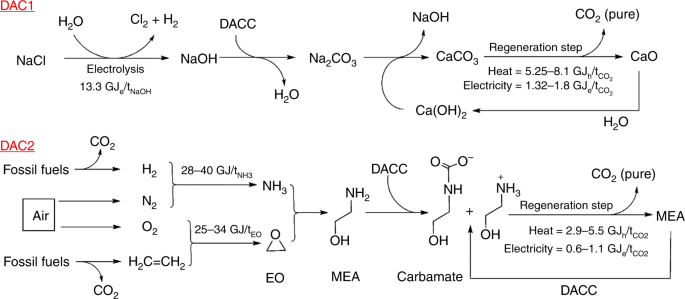
Unrealistic energy and materials requirement for direct air capture in deep mitigation pathways | Nature Communications
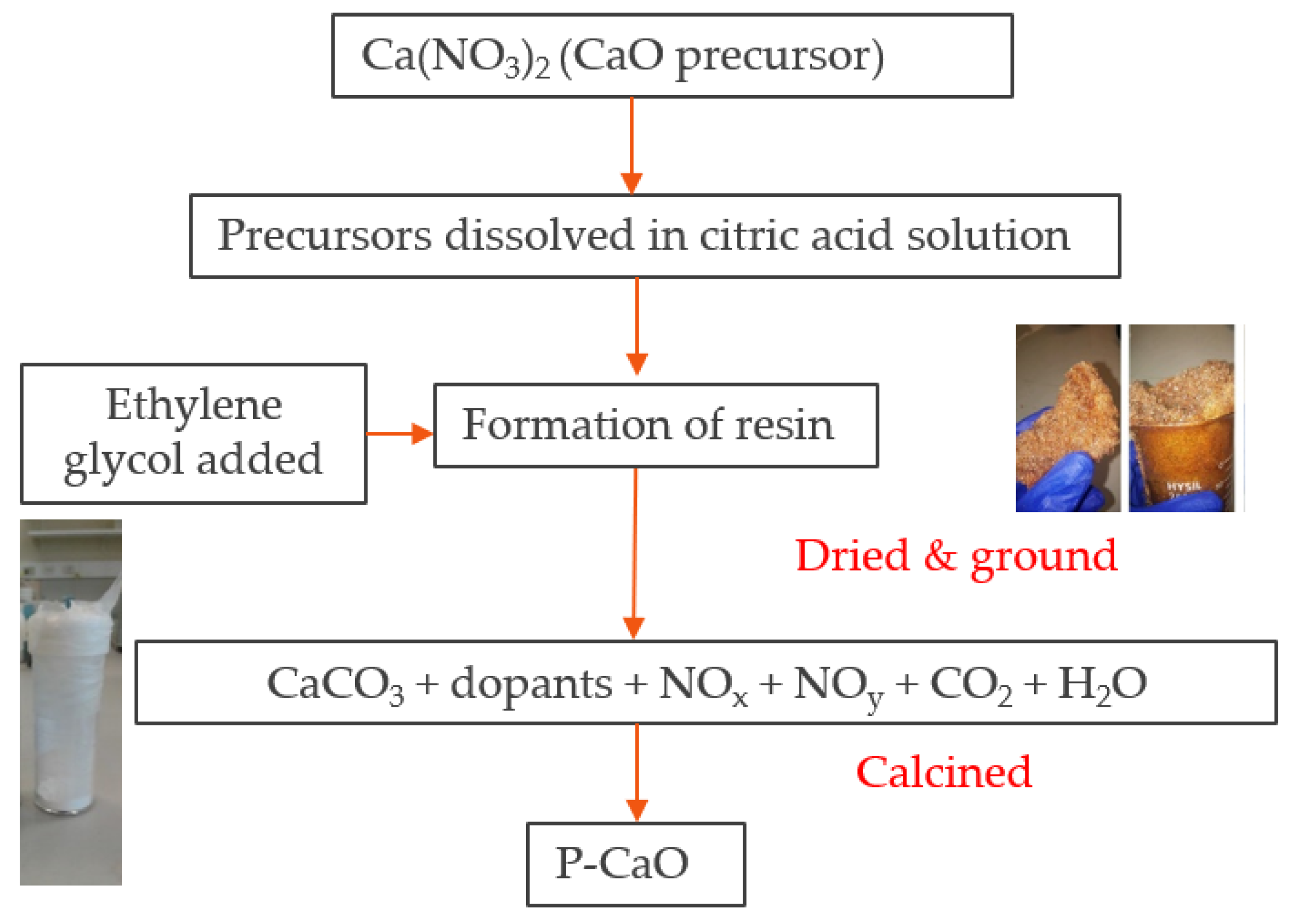
Applied Sciences | Free Full-Text | Comparative Kinetic Analysis of CaCO3/CaO Reaction System for Energy Storage and Carbon Capture
SOLVED: 00 M sodium carbonate IS needed lo prepare 250 0 mL ofa 0 125 M solution? What volume of 4 Attach File Browse My Computer @uesTion 10 Ifa 10.00 mL solution

Electrochemical Splitting of Calcium Carbonate to Increase Solution Alkalinity: Implications for Mitigation of Carbon Dioxide and Ocean Acidity | Environmental Science & Technology
SOLVED: You analyzed 2.475 g of a mixture of NaCl, SiO2, and CaCO3. You isolate 0.825 g NaCl, 0.825 g of CaCO3, and 0.580 g SiO2. You were told that the original

Polymorph Selection and Structure Evolution of CaCO3 Mesocrystals under Control of Poly(sodium 4-styrenesulfonate): Synergetic Effect of Temperature and Mixed Solvent | Crystal Growth & Design









