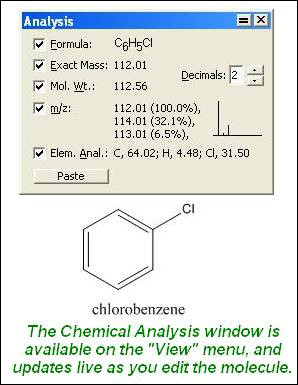
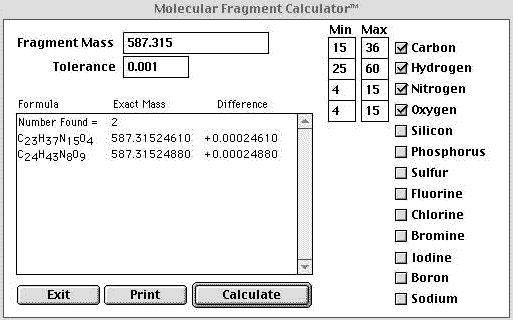
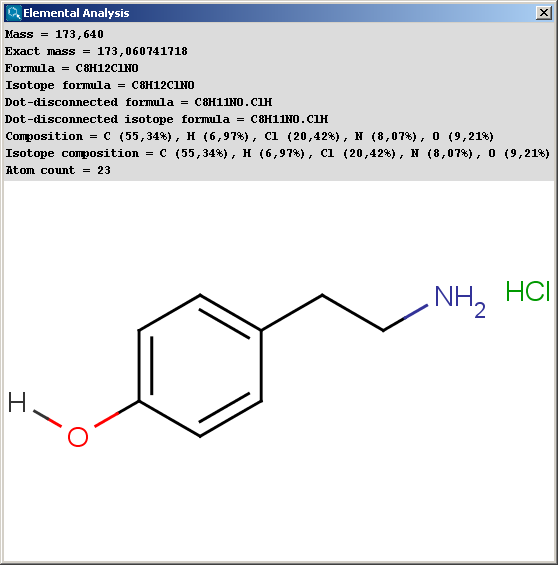
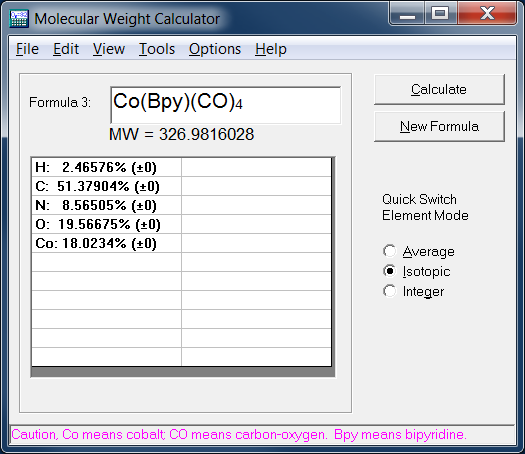
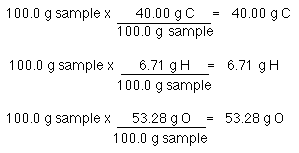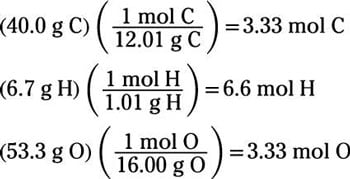SOLVED: compound is found to have the following elemental analysis: Carbon 65.02% Hydrogen 6.06 Oxygen 9.62 % Sulfur 19.29 % Calculate the empirical formula of this compound ts)

Question Video: Identifying the Chemical Formula of Oxalic Acid from Its Percent Composition of Constituent Elements | Nagwa