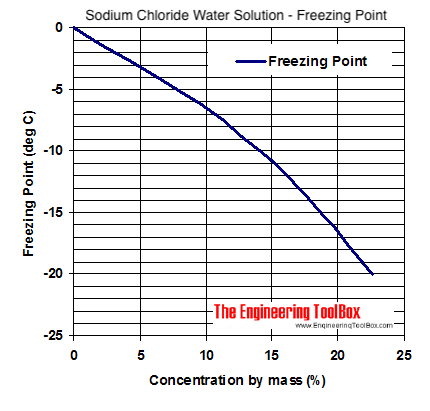
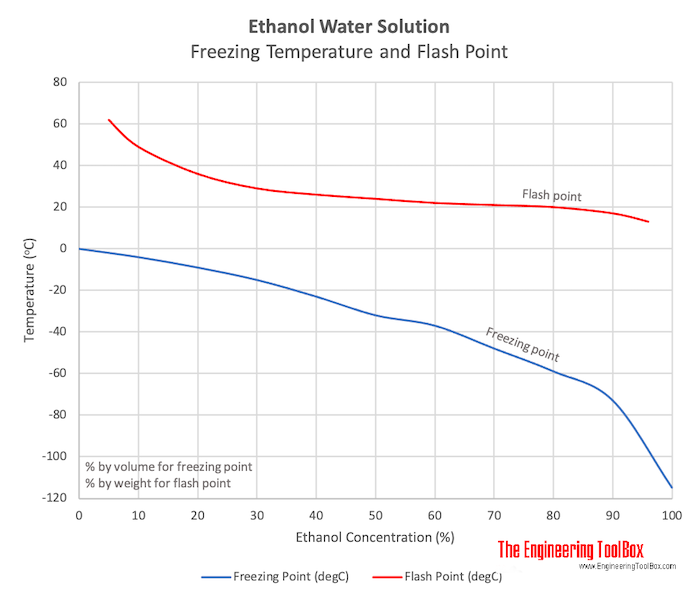
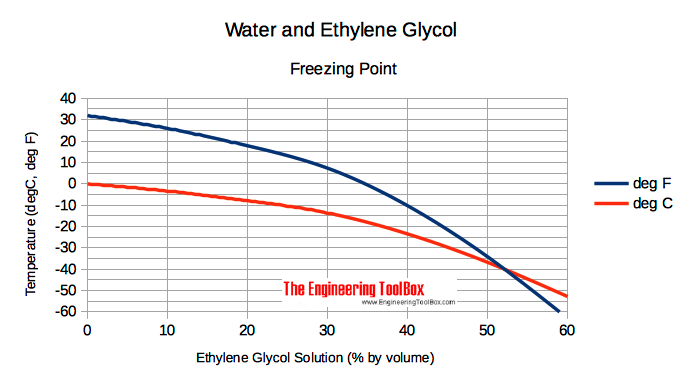
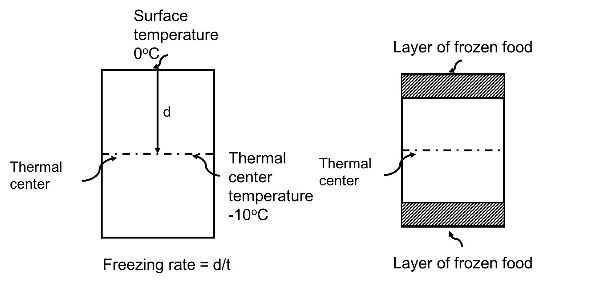
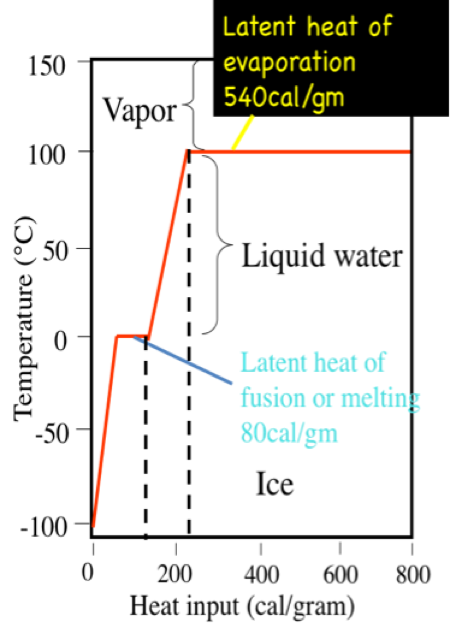
In an experiment, 17 g of ice is used to bring down the temperature of 40 g of water at 34^∘C to its freezing temperature . The specific heat capacity of water

Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube