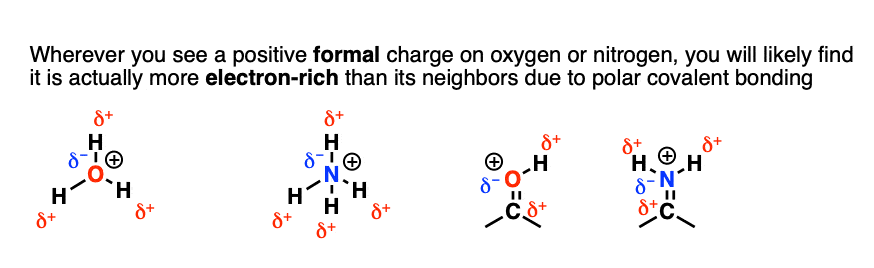Self-consistent calculations of charge self-trapping energies: A comparative study of polaron formation and migration in PbTiO3 | Computational Materials Group @ Chalmers
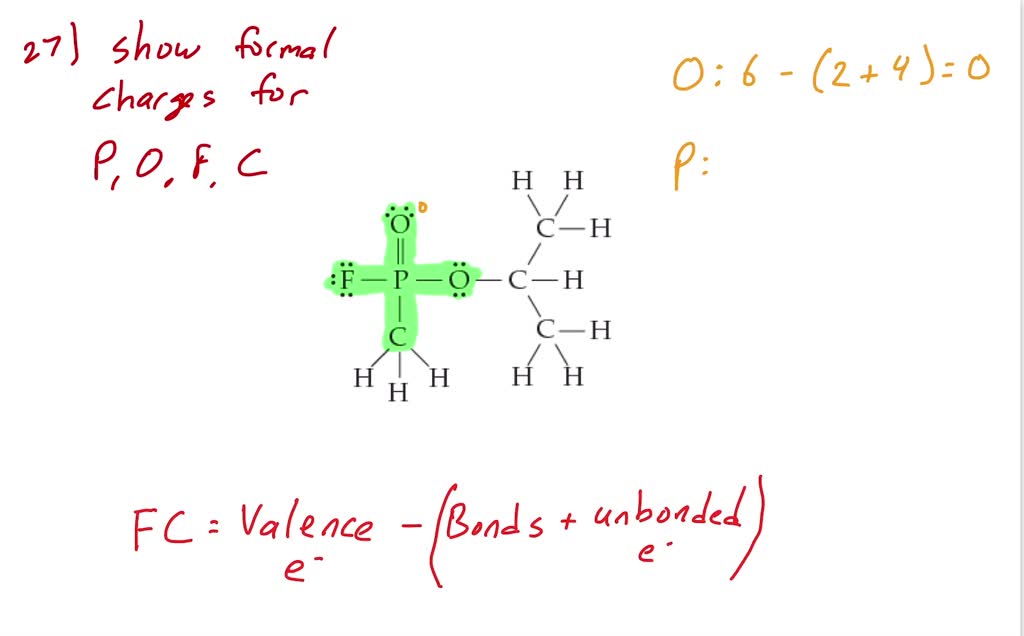
SOLVED: The electron-dot structure for the nerve agent, Sarin is shown. Calculate the formal charges on P and the atoms bonded to it F, C, and two O's.
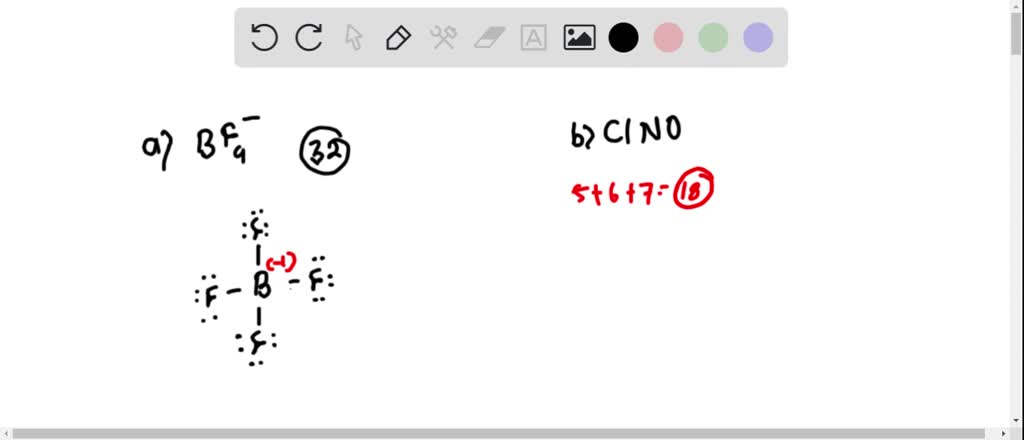
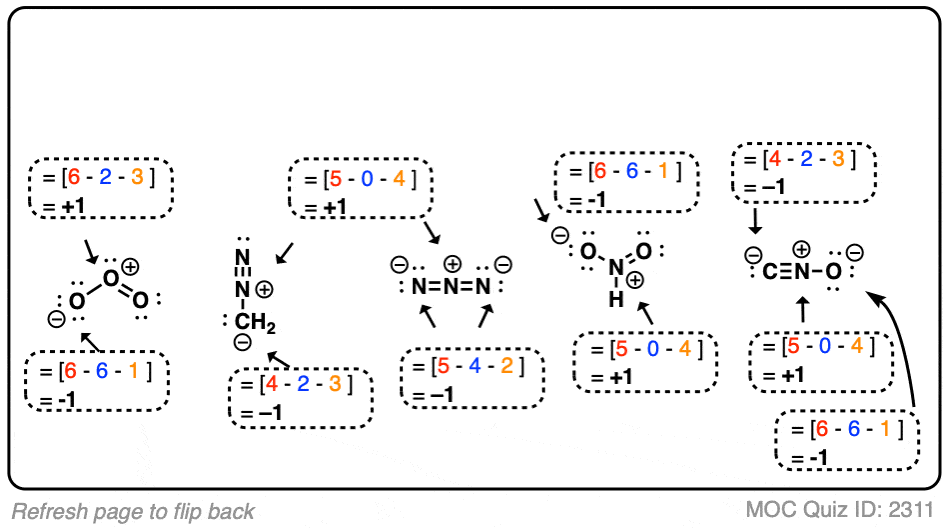
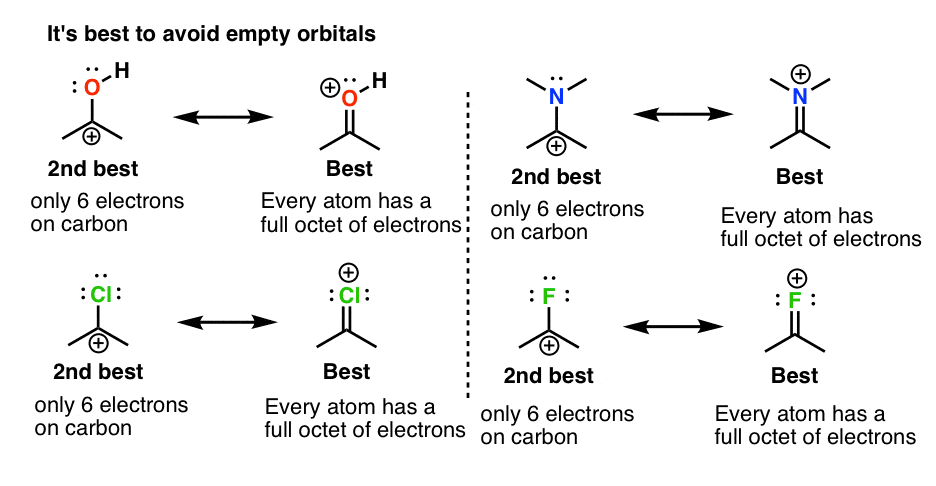
SOLVED: TALGDDAAOA Wel SCREEA CO Rareaa naso BACK NEXT COMPARING LEWIS STRUCTURES T02/502 Enter the overall charge for each substance For non zero charge greater than ont enter the number first and

:max_bytes(150000):strip_icc()/terms_e_ebit_FINAL-62447fe6ba024675aa01ade66cbaf109.jpg)




:max_bytes(150000):strip_icc()/COST-OF-CAPITAL-FINAL-HR-f3d41d21c66a494ea77eec360a6a3857.jpg)