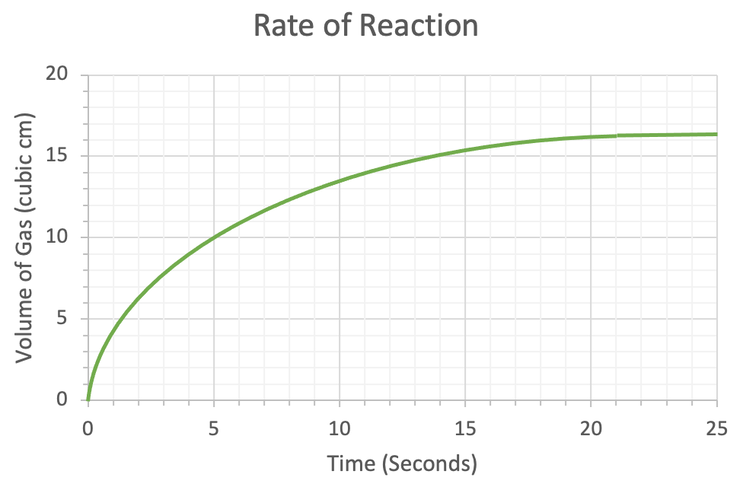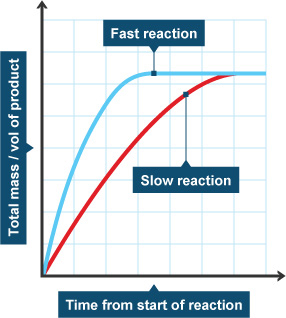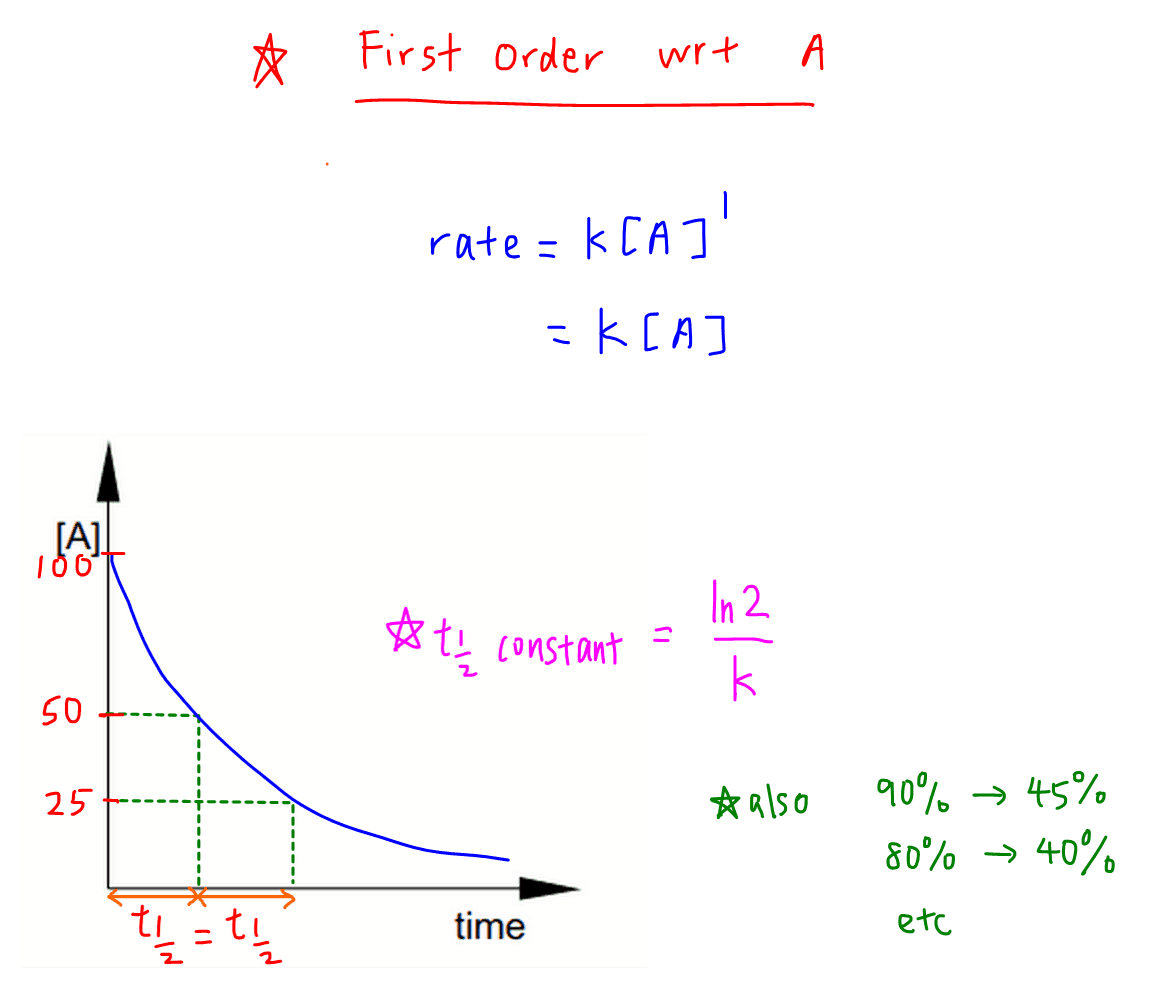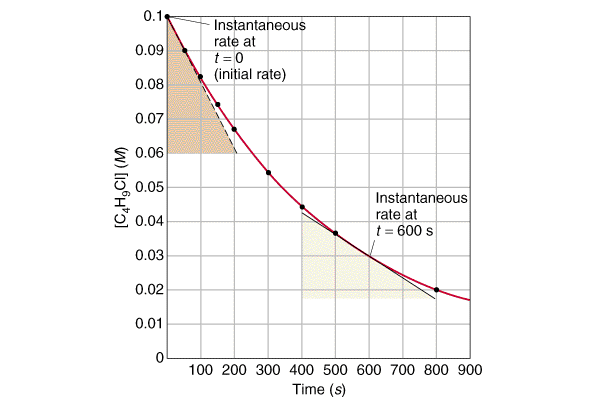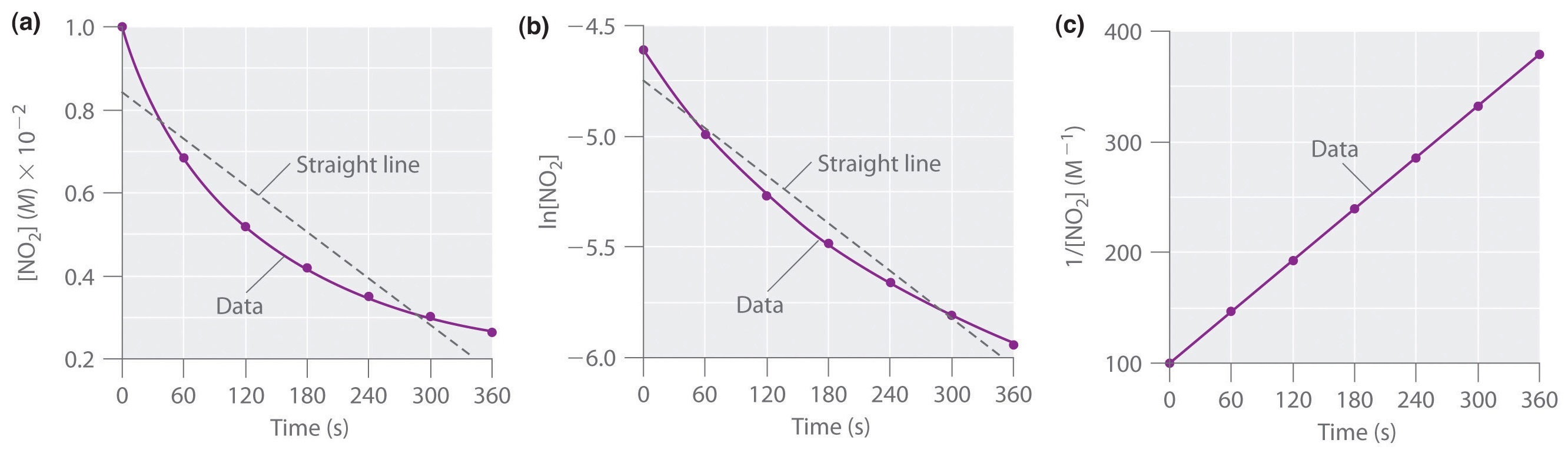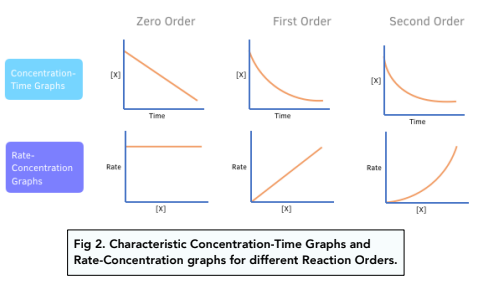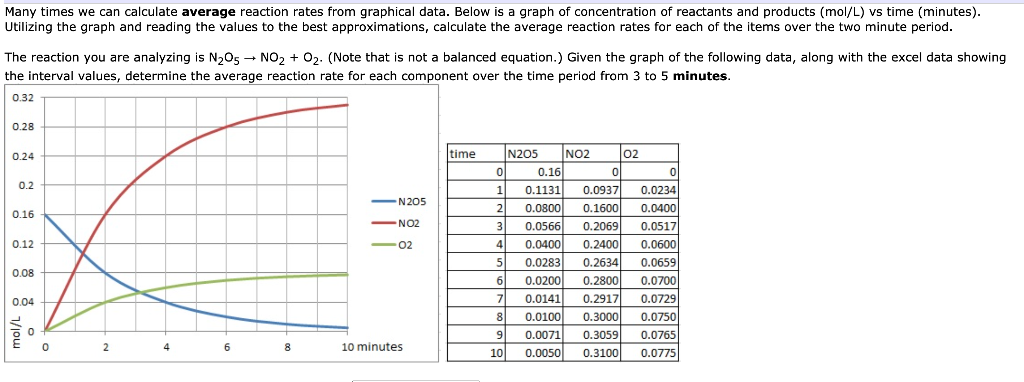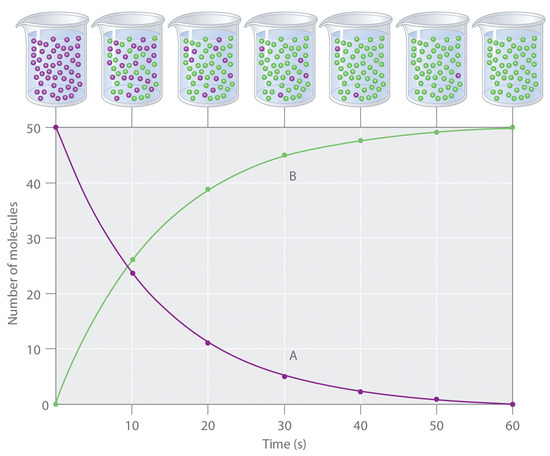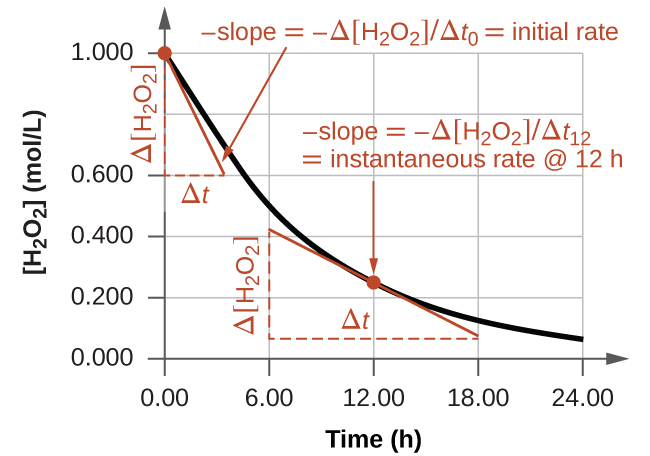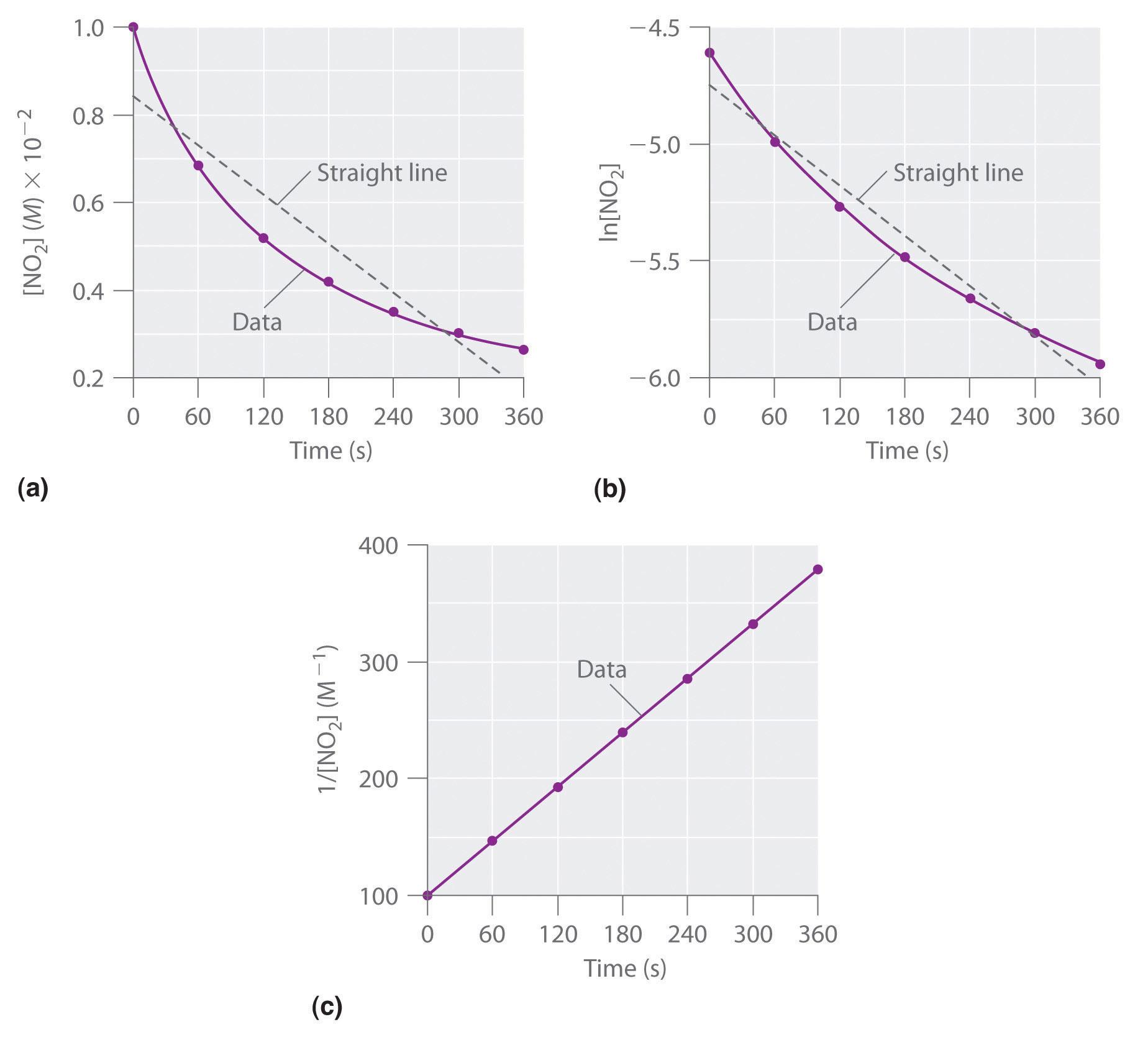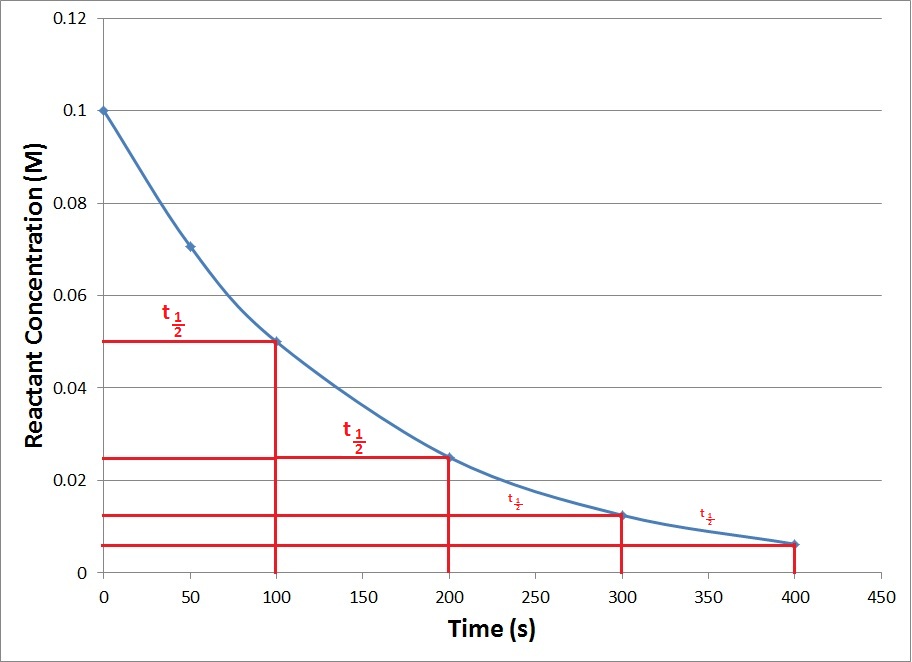
Concentration–Time Relationships: Integrated Rate Laws – Introductory Chemistry – 1st Canadian Edition

Calculating Average and Instantaneous Reaction Rate from a Graph of Concentration Versus Time | Chemistry | Study.com
How to determine rate of reactions|, chemical kinetics - High School/Honors/AP® Chemistry Resources | Viziscience
![The concentration of a compound A varies with time as follows: (a) Plot [A], ln [A], and [A]^{-1} versus time, and determine the order of the reaction with respect to [A]. (b) The concentration of a compound A varies with time as follows: (a) Plot [A], ln [A], and [A]^{-1} versus time, and determine the order of the reaction with respect to [A]. (b)](https://homework.study.com/cimages/multimages/16/zzz1098376891806932169.jpg)