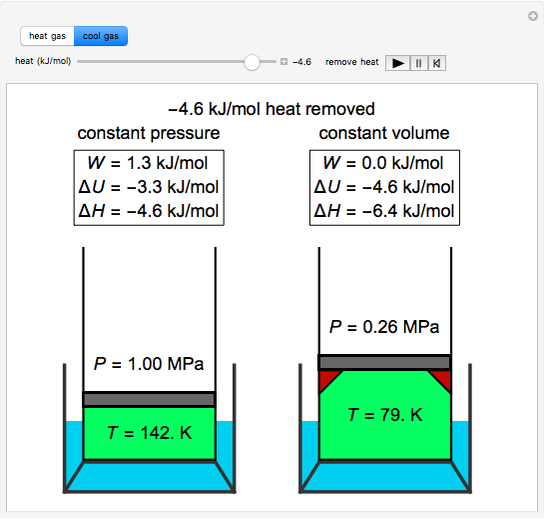
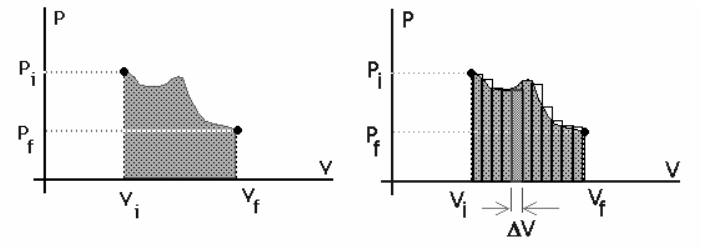
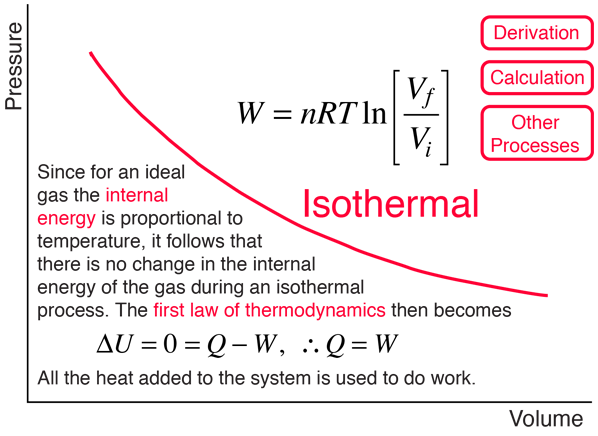
Fig. S 6: Gas expansion factor as a function of calculated centreline... | Download Scientific Diagram

A gas expands from `3 dm^(3)` to `5 dm^(3)` against a constant pressure of 3 atm. The work done - YouTube
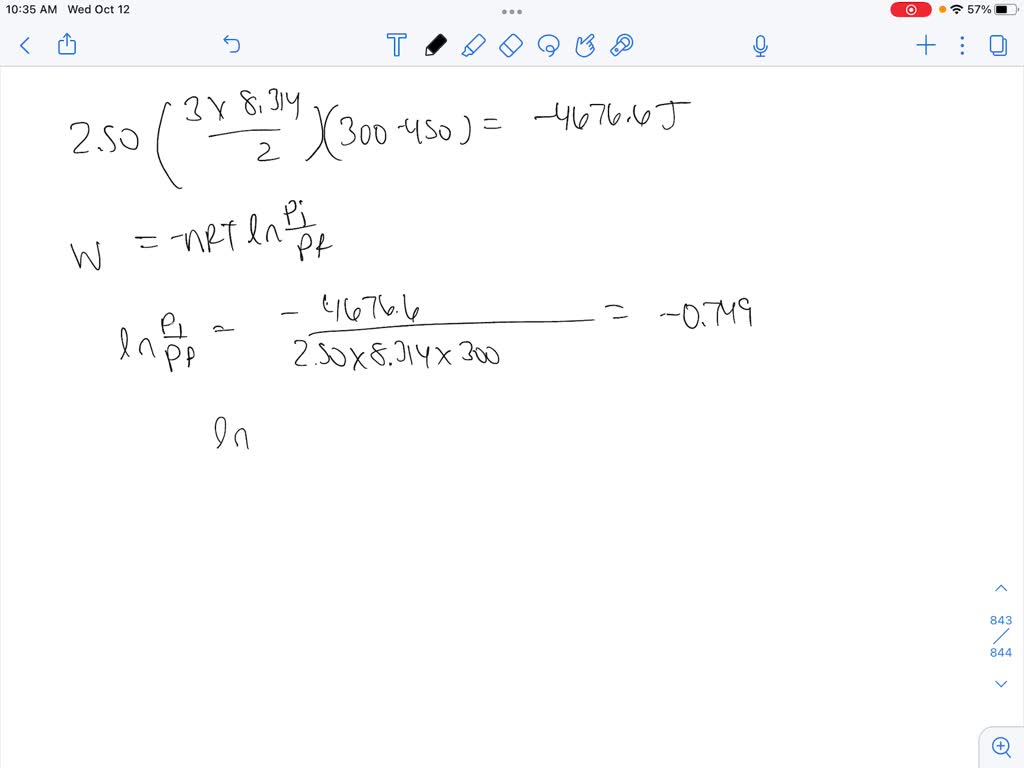
SOLVED: P4) Calculate w for the adiabatic expansion of 1 mol of an ideal gas at an initial pressure of 2.00 bar from an initial temperature of 450 K to a final