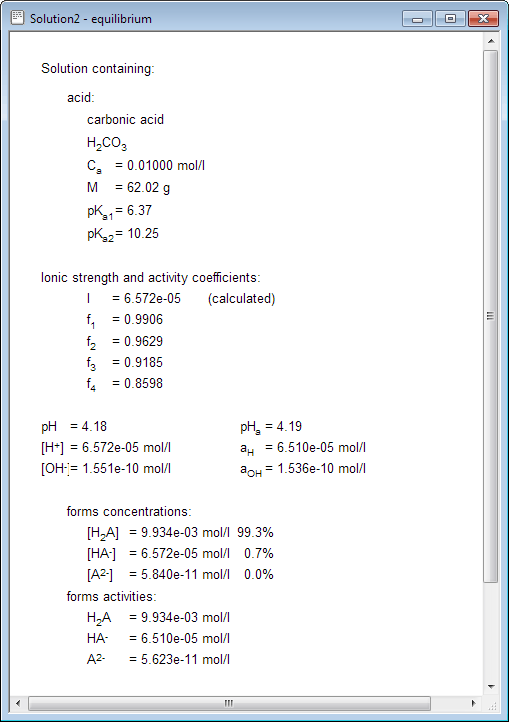
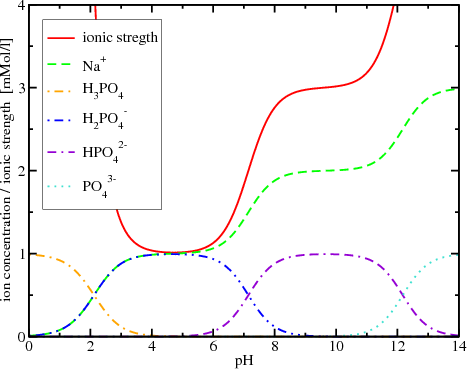
Example Write a charge balance equation for a solution containing KI and AlI3. Solution KI g K+ + I- AlI3 = Al I- H2O D H+ + OH- The equation can. -

Example Write a charge balance equation for a solution containing KI and AlI3. Solution KI g K+ + I- AlI3 = Al I- H2O D H+ + OH- The equation can. -