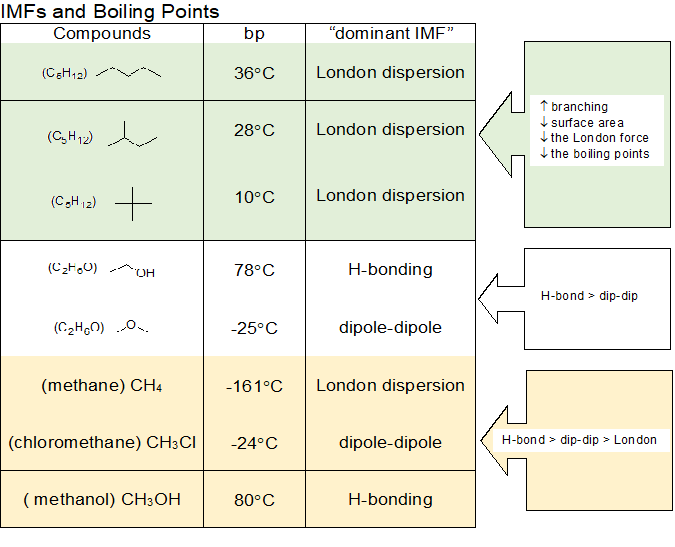
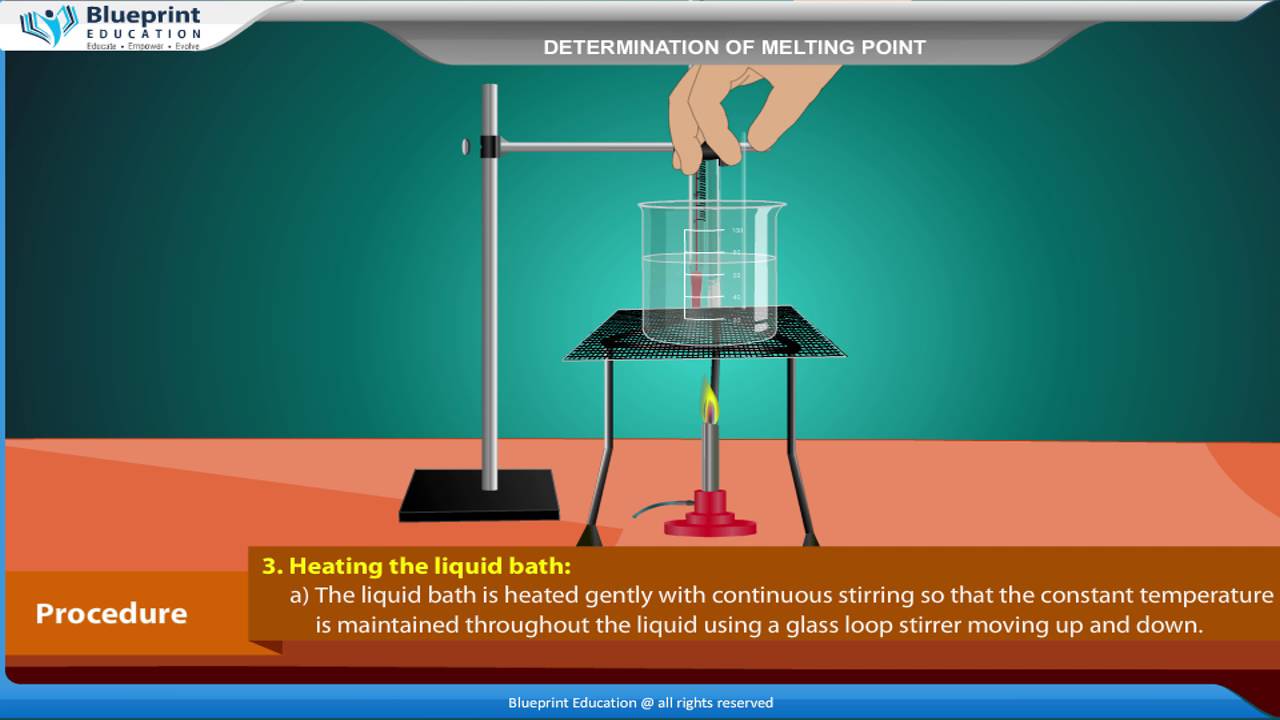
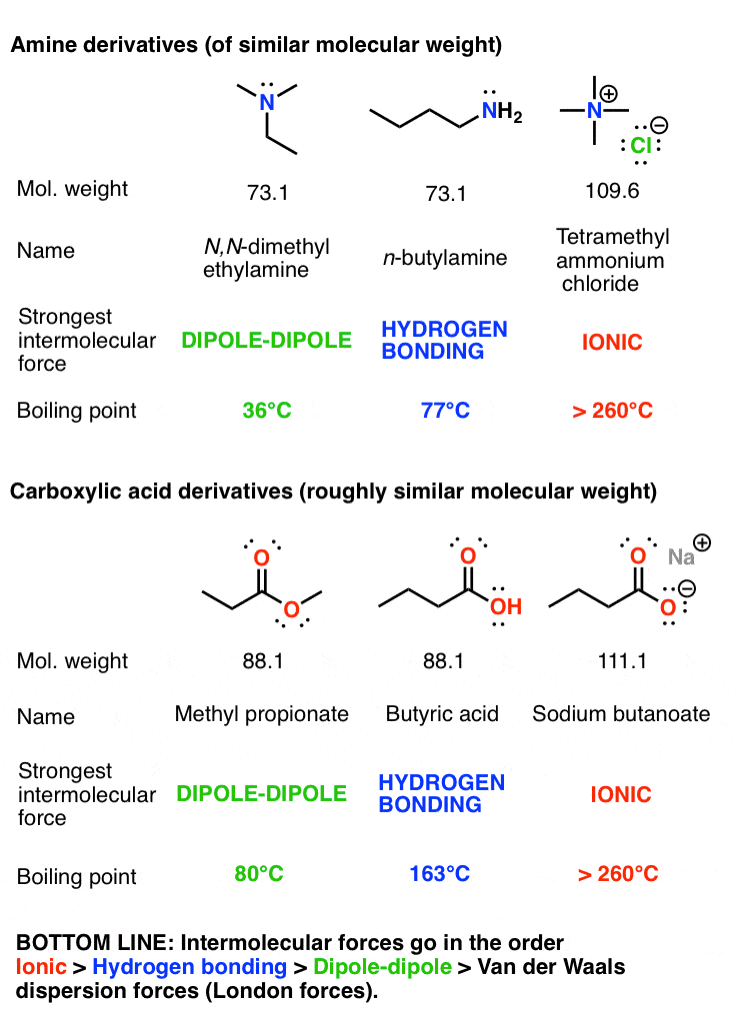
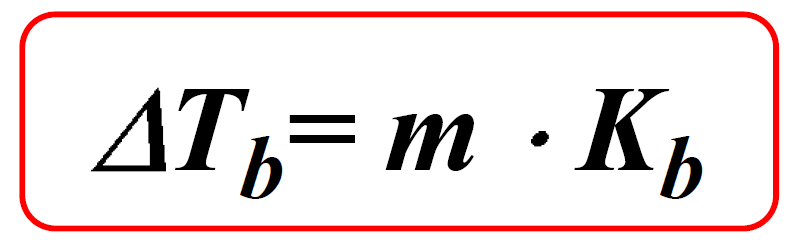Calculate the mass of ascorbic acid to be dissolved in 75 g acetic acid to lower its melting point by 1.5 ̊C , Kf = 3.9 K kg / mol – The Unconditional Guru

SOLVED: A rudimentary method to calculate the melting temperature of DNA is an equation known as "The Wallace Rule" This equation is: Tm = 2(A+T) + 4(G+C) Where A = # of