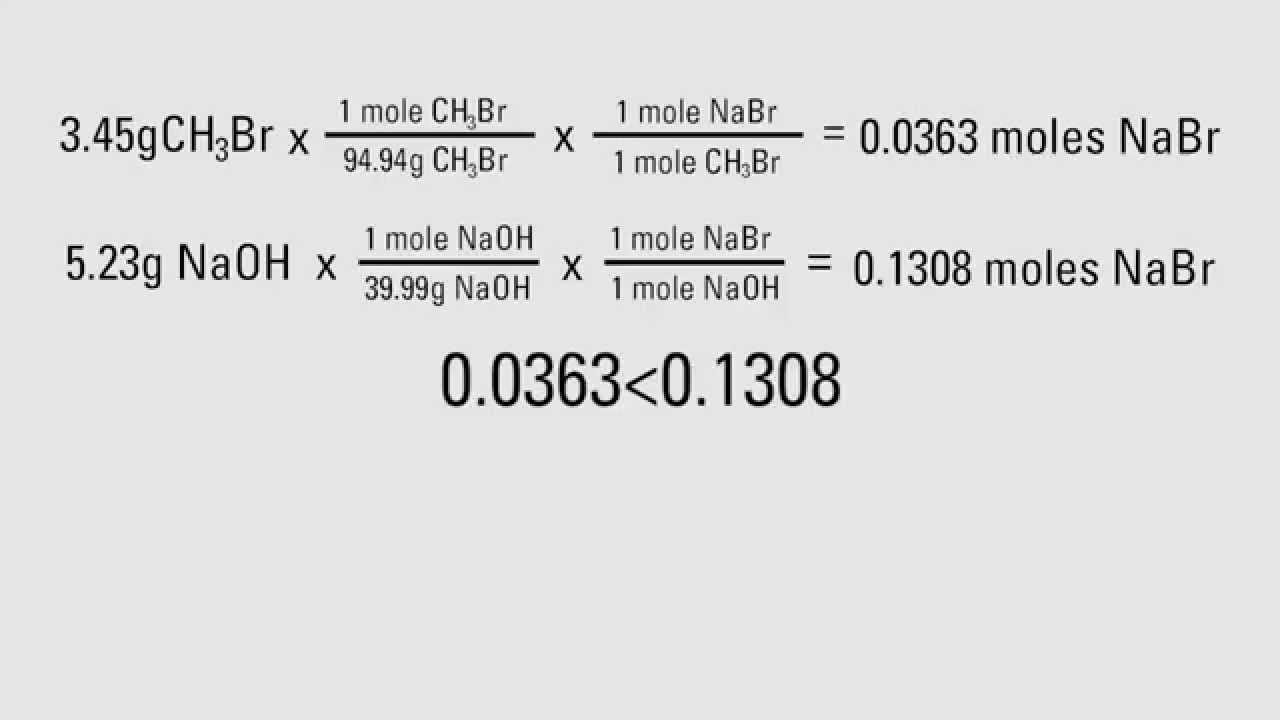
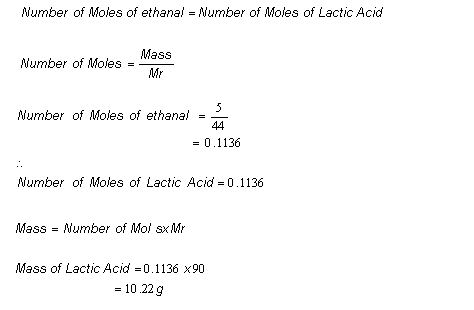8.6: Limiting Reactant, Theoretical Yield, and Percent Yield from Initial Masses of Reactants - Chemistry LibreTexts
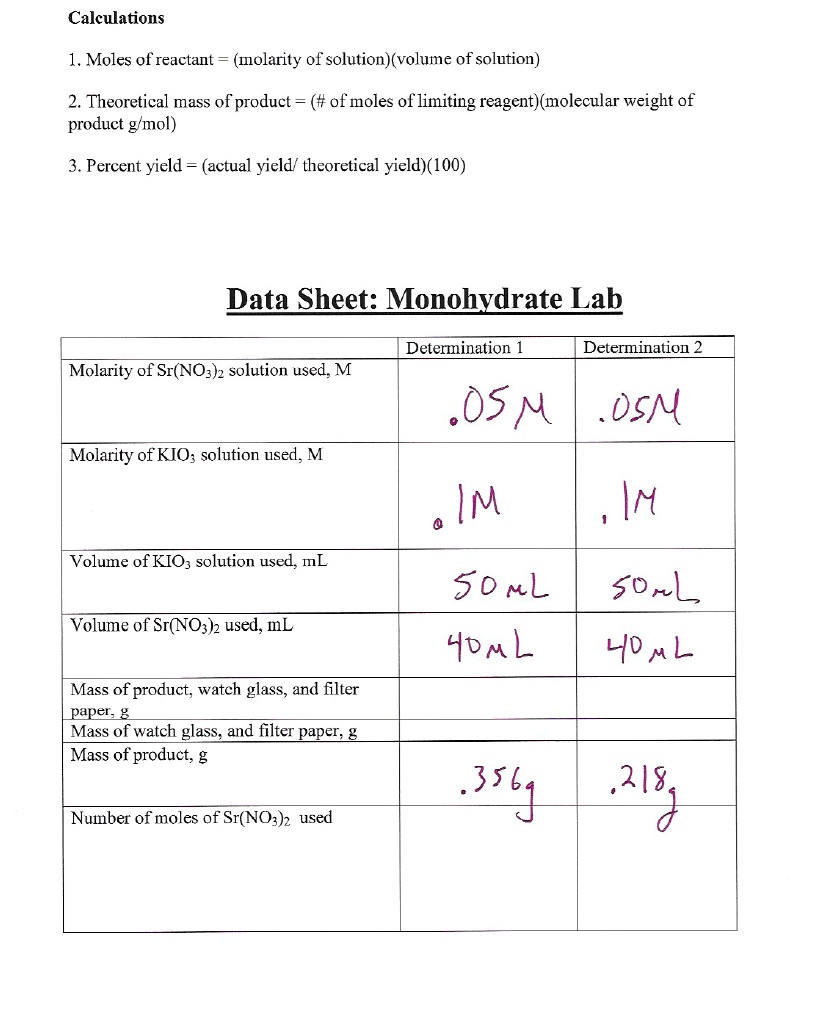
SOLVED: Calculations 1. Moles of reactant (molarity of solution)(volume of solution) 2. Theoretical mass of product (# of moles of limiting reagent)(molecular weight of product gmol) Percent yield :- (actual yield' theoretical

Theoretical, Actual, Percent Yield & Error - Limiting Reagent and Excess Reactant That Remains - YouTube

SOLVED: Actual Yield % Yield = Theoretical Yield 100 Mass Density = Volume (mL) Grams Mole = Molecular weight Moles M = Liter Moles = M X Liter

Question Video: Calculating the Percentage Yield of the Recreation of Aqueous Copper Sulfate with Zinc Metal | Nagwa
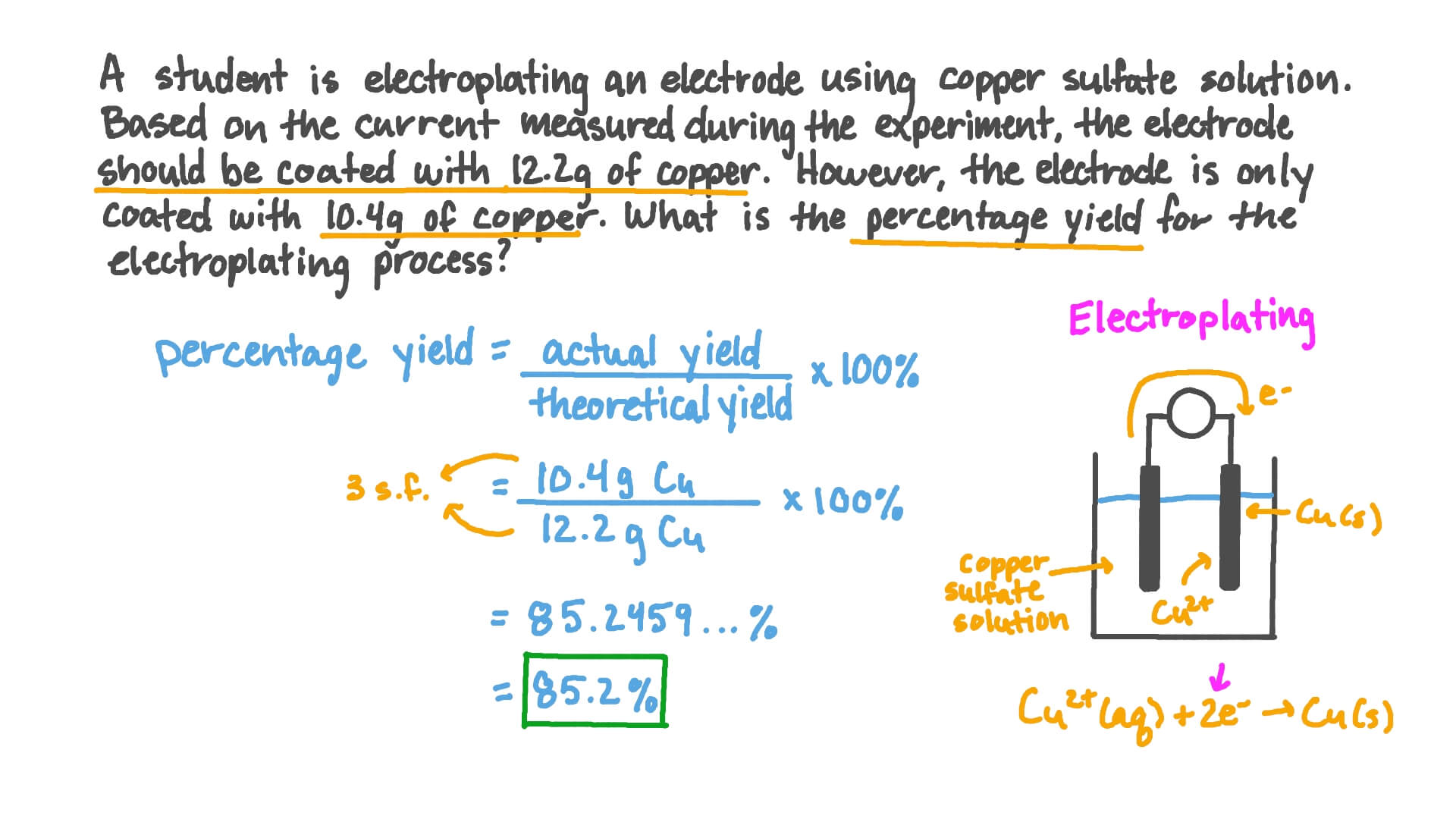
Question Video: Calculating the Percentage Yield from Given Actual Yield and Theoretical Yield | Nagwa