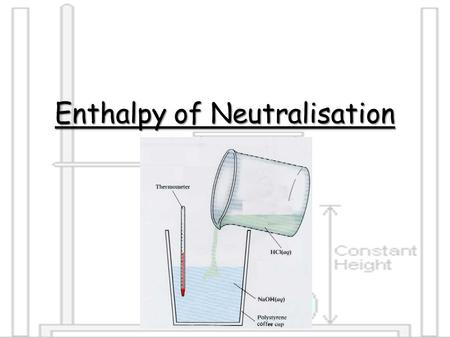
Aim: How can we describe the neutralization reaction? Do Now: 1.Take out a calculator and reference tables. 2.Which of the following will react with an. - ppt download

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa

SOLVED: Table 3: Heat of Neutralization (HC,H,0,as acid) Show calculations scparately: Temp of calorimeter and NaOH before mixing ("C) Temp of HCH,O, before nixing ("C) Z2 230 Final Temperature extrapolated from graph ("

SOLVED: ar Enthalpy or Reaction Make these assumptions, treating the aqueous solutions as if they were pure water: Calorimeter constant determined Part Or , skipped, considered 0 determine using temperature and Appendix

Gibb's free energy calculation result of neutralization reaction of H 2... | Download Scientific Diagram


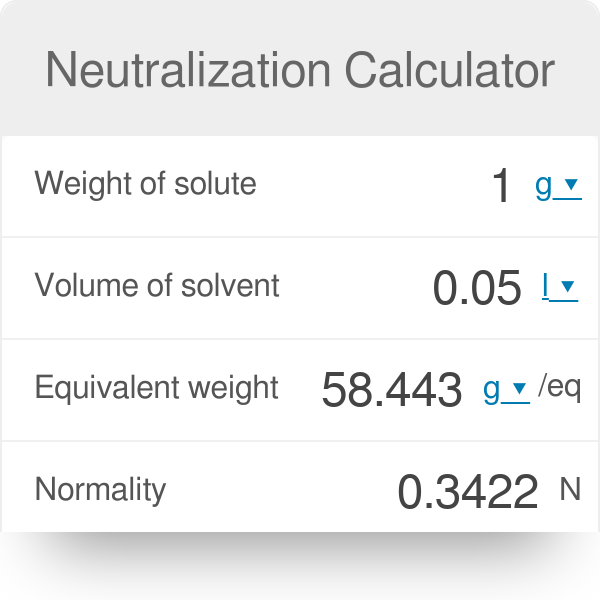




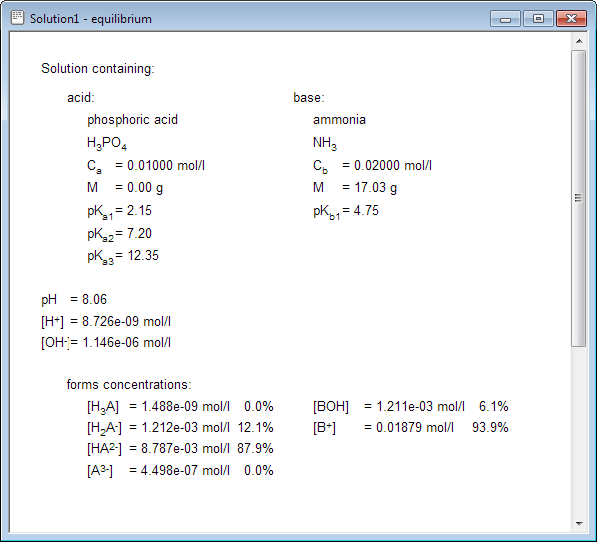
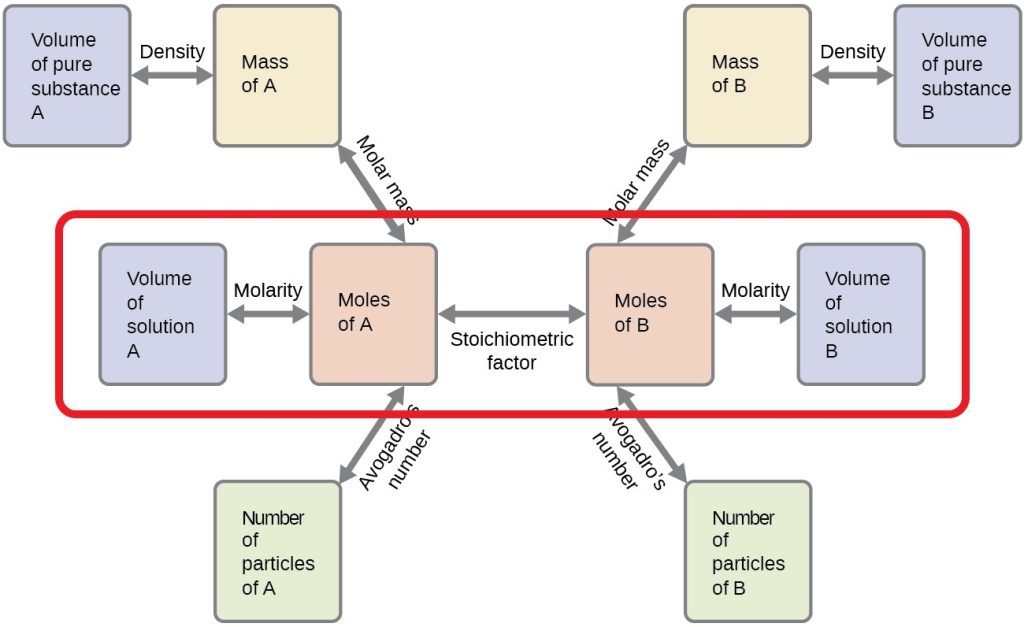


![4.4] Heat of neutralisation - Calculation - YouTube 4.4] Heat of neutralisation - Calculation - YouTube](https://i.ytimg.com/vi/x_993x_cBh8/hqdefault.jpg)



