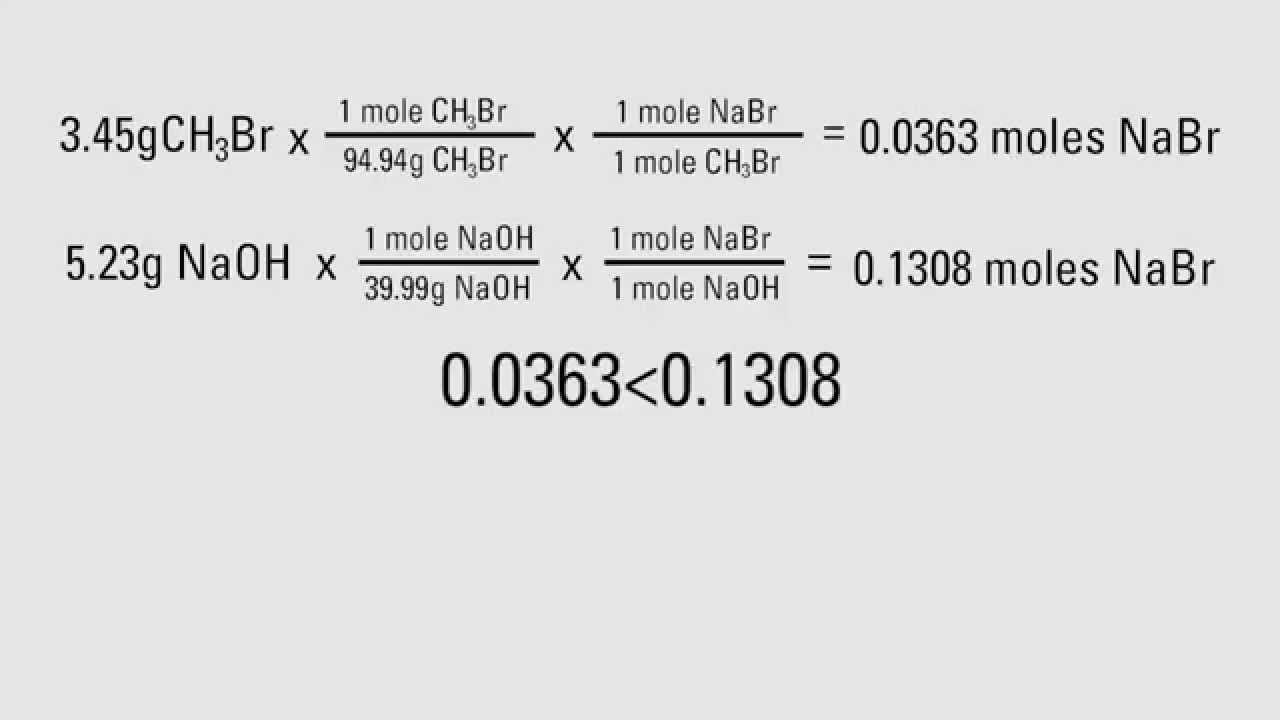
8.6: Limiting Reactant, Theoretical Yield, and Percent Yield from Initial Masses of Reactants - Chemistry LibreTexts

How to Calculate Theoretical Yield - Definition and Example | Chemical equation, Molecular mass, Chemical reactions

SOLVED: Calculate the percentage yield of Benzil. Made from 4g of Benzoin using 14mL of concentrated nitric acid. Initial mass of Benzil after vacuum filtration: 4.593g Mass after drying: 3.388g