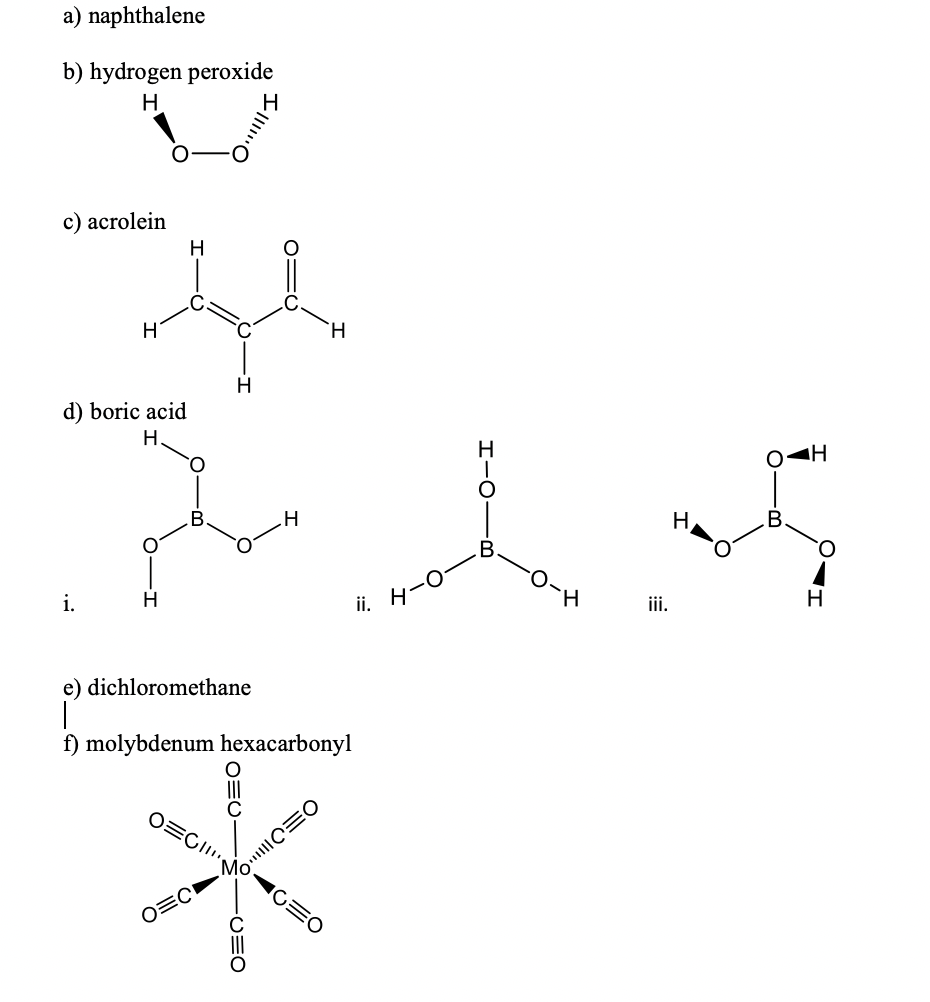
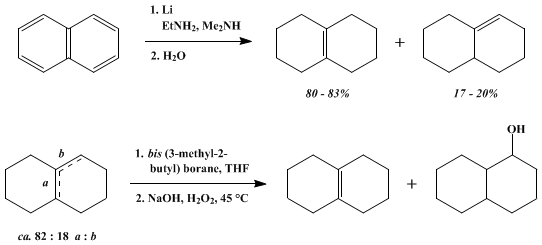
4) 120 ML · Hydrogen peroxide is manufactured bye auto - oxidation of : (1) 2 - ethyl - anthraquinol (2) antraquinone (3) naphthalene (4) anthracene and DO

Hydrogen peroxide is manufactured by the auto-oxidation of: (A) 2-ethyl-anthraquinol (B) anthraq... - YouTube

Comparison of naphthalene removal performance using H2O2, sodium percarbonate and calcium peroxide oxidants activated by ferrous ions and degradation mechanism - ScienceDirect

Oxidation of polycyclic aromatic hydrocarbons with hydrogen peroxide catalyzed by Iron(III)porphyrins - ScienceDirect
![PDF] Unexpected cyanosis in a haemodialysis patient—did someone add hydrogen peroxide to the dialysis water? | Semantic Scholar PDF] Unexpected cyanosis in a haemodialysis patient—did someone add hydrogen peroxide to the dialysis water? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6ab2946a8f759f75ee87daef3afb7b9f199adf34/2-Table2-1.png)
PDF] Unexpected cyanosis in a haemodialysis patient—did someone add hydrogen peroxide to the dialysis water? | Semantic Scholar
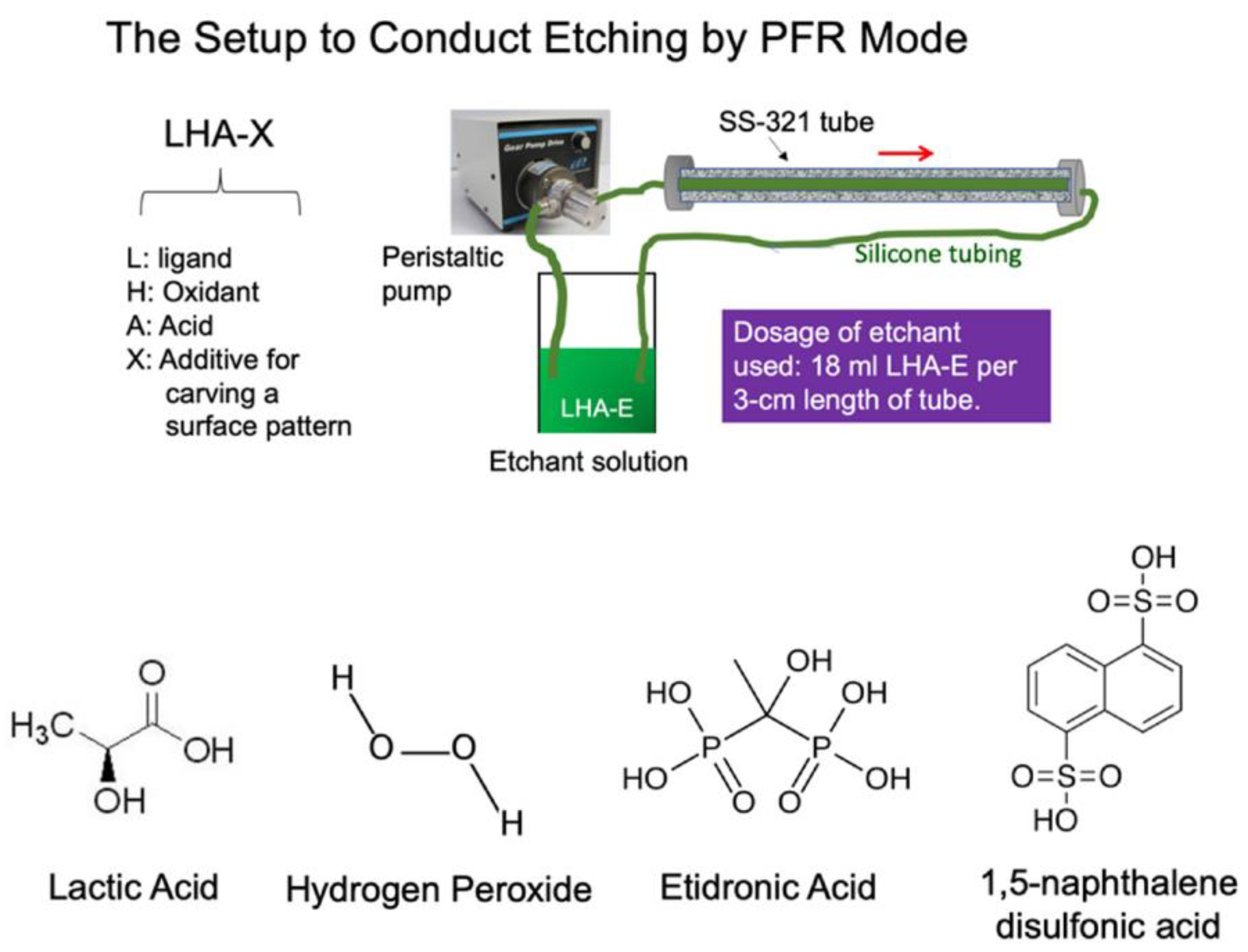
J. Compos. Sci. | Free Full-Text | Crafting Metal Surface Morphology to Prevent Formation of the Carbon–Steel Interfacial Composite

Naphthalene – Sulfuric Acid,hydrochloric acid,nitric acid,hydrogen peroxide,glacial acetic acid,formic acid,caustic soda supplier
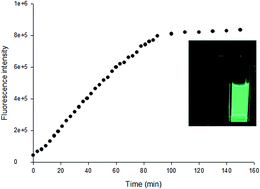
A novel two-photon fluorescent probe for the selective detection of hydrogen peroxide based on a naphthalene derivative - Analytical Methods (RSC Publishing)

Reduction of hydrogen peroxide by naphthalene peri-diselenide-based glutathione peroxidase mimics: Mechanism and effect of substitutions - ScienceDirect

Selective oxidation of naphthalene derivatives with ruthenium catalysts using hydrogen peroxide as terminal oxidant - ScienceDirect

Reduction of hydrogen peroxide by naphthalene peri-diselenide-based glutathione peroxidase mimics: Mechanism and effect of substitutions - ScienceDirect

Hydrogen Peroxide-coupled cis-Diol Formation Catalyzed by Naphthalene 1,2-Dioxygenase - ScienceDirect
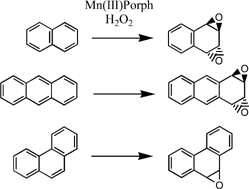
An efficient approach for aromatic epoxidation using hydrogen peroxide and Mn(iii) porphyrins - Chemical Communications (RSC Publishing)